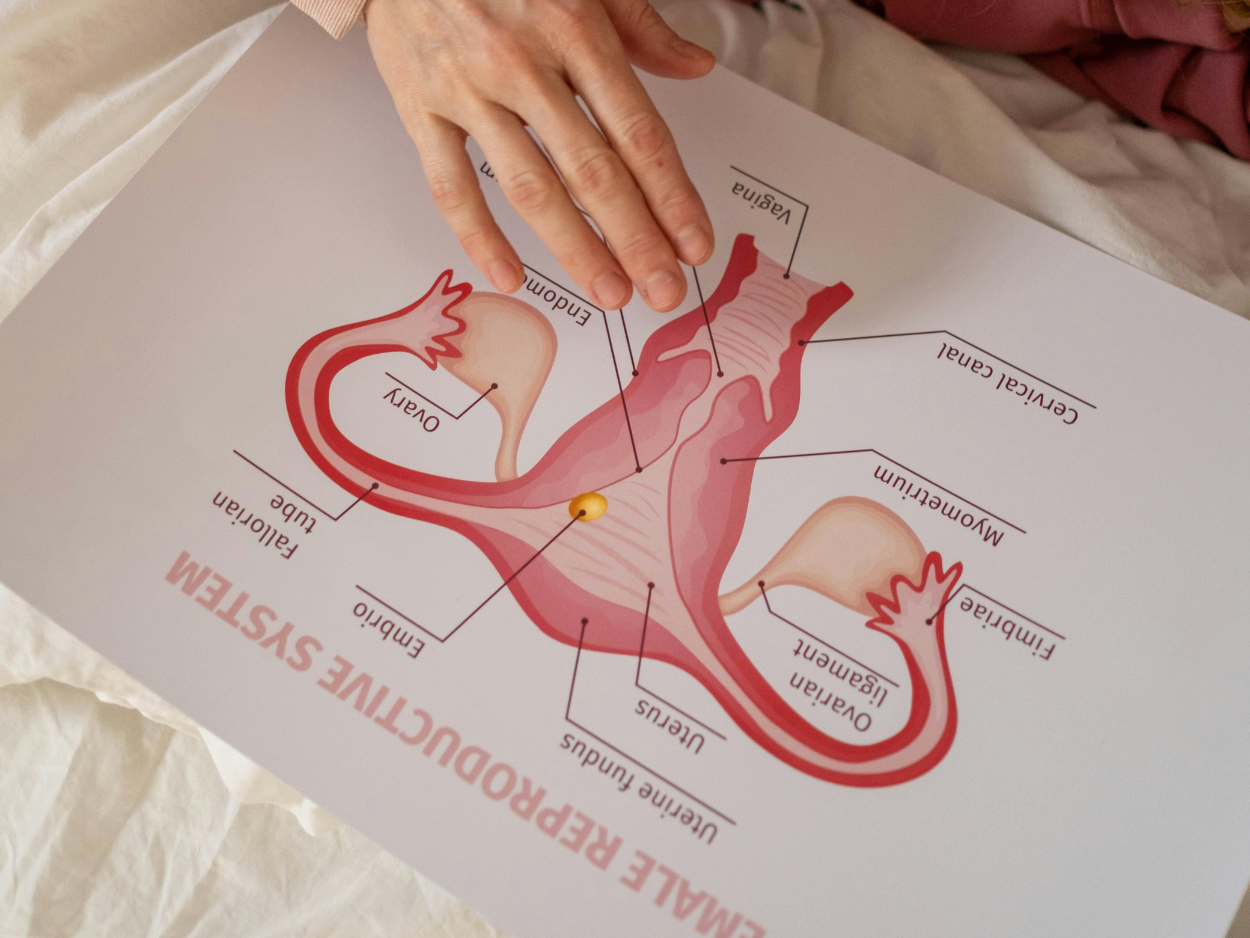
Photobiomodulation's role in reproductive health is one of the most promising research areas in the field. Barbara Sekulovska shares what the science — and our clinical partnership — is beginning to show.
Table of Contents
- Why Fertility Became Part of Our Research
- What the Science Already Tells Us
- How Red Light Therapy Affects Oozytes
- Our Clinical Partnership: How It Started
- What We Are — and Aren't — Claiming
- The Kini Fertility Protocol
- Frequently Asked Questions
- Who This Article Is For
I want to start this article the way I wish someone had started a conversation with me a few years ago - honestly, and without overselling.
Fertility is one of the most emotionally loaded health topics there is. If you're reading this because you're actively trying to conceive, or supporting someone who is, I want to be careful with your attention. I'm not going to tell you that red light therapy for fertility is a miracle. I'm going to tell you what the research shows, what we're beginning to observe in a clinical setting, and where I think this is genuinely going.
That's it. You can decide what to do with it.
1. Why Fertility Became Part of Our Research
When Thomas and I started building Luminous Labs, our initial focus was recovery - athletic performance, sleep, skin. The connection to reproductive health emerged from our customers, not from our roadmap.
We started hearing from women who were going through IVF, dealing with endometriosis, managing PCOS, or simply trying to conceive in their late thirties. They weren't asking us about athletic recovery. They were asking whether the same cellular mechanism that we talked about for energy and repair had any relevance to reproductive health.
The honest answer was: yes, and more than we initially appreciated.
Reproductive cells - particularly oocytes - are among the most metabolically demanding cells in the human body. Their function depends heavily on a molecule called ATP. And ATP production, as you may know from our previous writing, is exactly where photobiomodulation has its most well-documented effect.
That observation led us to a deeper literature review, and eventually to a conversation with one of Austria's most respected fertility clinics.
2. What the Science Already Tells Us
The research on photobiomodulation and reproductive health is not brand new - but it is underreported.
The foundational piece is mitochondrial function. A landmark review by Bentov and Casper (2013), published in Fertility and Sterility, established that mitochondrial dysfunction in oocytes is a primary driver of age-related decline in egg quality. ¹ The study found that as mitochondrial ATP output decreases, so does fertilisation rate, embryo development quality, and implantation success. The authors concluded that interventions capable of improving mitochondrial function in oocytes represented a meaningful clinical opportunity.
Enter photobiomodulation. A 2016 clinical study by Kinoshita et al., published in Laser Therapy, applied low-level laser therapy (LLLT) to poor-prognosis IVF patients - women who had previously failed multiple IVF cycles. The results showed statistically significant improvements in fertilisation rates and embryo quality in the treated group compared to controls. ² The researchers attributed the effect to increased mitochondrial activity in the oocytes following light exposure.
The mechanism makes sense when you understand it: red and near-infrared light at the right wavelengths activates cytochrome c oxidase, the terminal enzyme in the mitochondrial electron transport chain. ³ This drives increased ATP synthesis and reduces reactive oxygen species - the oxidative stress that has been implicated in poor oocyte quality, failed implantation, and early pregnancy loss. ⁴
None of this is fringe science. It is photochemistry applied to one of the most ATP-dependent processes in human biology.
3. How Red Light Therapy Affects Oozytes
Oocytes - human egg cells - contain more mitochondria than almost any other cell type in the body. This is not incidental. It reflects the extraordinary energy demands of fertilisation, early cell division, and embryo development.
When mitochondrial function in an oocyte is compromised - whether through age, oxidative stress, or systemic cellular energy deficits - the downstream effects are well-documented: reduced fertilisation competence, poor embryo quality, chromosomal instability, and lower implantation rates.
Red light therapy at 630–660nm and 810–850nm works on the mitochondria in oocytes through the same cytochrome c oxidase pathway that underpins all photobiomodulation effects. When these wavelengths are absorbed by the mitochondrial chromophore, ATP synthesis accelerates, reactive oxygen species decrease, and cellular repair signalling is upregulated. ³
What makes oozytes particularly interesting in this context is their accessibility. Unlike muscle cells deep in tissue, the ovarian environment can be reached with appropriate near-infrared wavelengths delivered externally. This is a key reason why clinical interest in LLLT for reproductive medicine has grown steadily over the past decade.
For the uterine environment, the anti-inflammatory and pro-vascular effects of photobiomodulation are also relevant — improved local circulation and reduced inflammatory cytokines may support implantation and early pregnancy. ²

4. Our Clinical Partnership: How It Started
About a year ago, I reached out to a colleague at one of Austria's most respected fertility clinics - Kinderwunschinstitut Dr. Schenk - to have an honest conversation about what the existing research showed and whether they would consider a structured observation protocol using the Kini and Essential.
What I appreciated about that conversation was the clinical rigour they brought to it immediately. They were not interested in anecdote. They wanted a defined protocol, clear documentation criteria, and absolute transparency about what we could and couldn't claim.
That is the partnership we built.
The protocol we are running is not a randomised controlled trial - we want to be clear about that. It is a structured observation: a defined cohort of patients using the Kini and/or Essential alongside their existing fertility treatment and/or in preparation for it, with standardised outcome tracking across a set of reproductive health markers.
We are not replacing any medical intervention. We are not adjusting anyone's medication or IVF protocol. We are adding a photobiomodulation component and documenting what we observe.
5. What We Are - and Aren't - Claiming
I want to be precise here, because this matters enormously.
We are not claiming that photobiomodulation treats infertility. These are early clinical observations — preliminary, hypothesis-generating, and not designed to assess pregnancy or live-birth outcomes. No causal conclusions can be drawn from cases of this scale. What I can share is what was observed, documented by treating clinicians, and reported in our internal white paper published January 2026.
Case 1: IVF Adjunct Use, Switzerland (FertiSuisse, n = 1)
A 39-year-old patient with six years of unsuccessful IVF cycles integrated PBM into an ongoing IVF protocol. PBM was applied via abdominal exposure - 20-minute sessions, three times per week.
Endometrial thickness was measured across multiple cycles, with and without PBM exposure. The pattern was striking: thickness increased during PBM use and partially regressed during non-use - a reversibility signal suggesting the effect is light-dependent rather than incidental.
Case 2: PBM Monotherapy, Austria (Kinderwunschinstitut Dr. Schenk, n = 12)
Following the Swiss case, an observational pilot was initiated at Kinderwunschinstitut Dr. Schenk, a respected Austrian fertility clinic. PBM was applied as a standalone intervention, without concurrent fertility medication. The cohort included women with chronically thin endometrium - a group that often shows limited responsiveness to standard hormonal approaches.
Endometrial thickness increased following PBM exposure across all documented cases with paired measurements. Several patients transitioned from values below commonly used clinical thresholds to values permitting embryo transfer or continuation of fertility treatment.
We were able to document an increase in endometrial thickness of over 100%. We also observed a higher oocyte yield following red light use compared to a prior cycle without it — this finding is exploratory and cannot be interpreted causally. The endomeitrum is influenced by a number of factors. At the time of reporting, one pregnancy had been clinically confirmed. No adverse events were reported across the cohort.
These observations are preliminary. They come from small, non-controlled cohorts. They do not prove efficacy. What they do - consistently, across two independent clinical settings - is suggest a reproducible signal in a patient population that standard approaches often fail.
That signal is what justifies the next phase of work: expanded cohorts, harmonised protocols, and prospective evaluation. The work is ongoing. We will share it as it develops.
6. The Fertility Protocol
Our products was not designed exclusively for fertility. But several of its design decisions make it particularly well-suited for this application.
Its wearable form factor allows targeted, hands-free use over the lower abdomen - the relevant treatment area for uterine and ovarian support. Its dual-wavelength output (630nm + 850nm) covers both the surface cellular layer and deeper tissue penetration. Its irradiance is calibrated to deliver a therapeutic dose without requiring prolonged sessions.
The protocol we suggest for women using our products in a fertility context - always alongside, never replacing, their medical care - is:
Daily use: 20 minutes over the lower abdomen, ideally at the same time each day for circadian consistency. Many users integrate this into a morning or evening routine.
Cycle awareness: Some users and clinicians prefer to pause use during the luteal phase if they are undergoing IVF embryo transfer, out of an abundance of caution while more data is gathered. We recommend discussing your specific cycle and treatment with your doctor.
Supporting use: Any of our products can also be used on the lower back and neck - for sleep support, stress regulation, and general hormonal balance - as complementary areas.
This is not a replacement for medical treatment. It is a cellular support tool - one that, based on the existing evidence and our ongoing observations, appears to be a meaningful addition to a comprehensive approach to reproductive health.
See how red light supports your goals - explore the product benefits now.

7. Frequently Asked Questions
Is red light therapy safe during IVF? The existing clinical literature, including the Kinoshita et al. study, has used LLLT within IVF protocols without adverse effects. However, every fertility treatment plan is individual. We strongly recommend discussing any supplementary protocol - including photobiomodulation - with your treating physician before beginning. Some clinicians prefer to pause use during specific phases of the IVF cycle.
Can red light therapy help with PCOS or endometriosis? The anti-inflammatory and mitochondrial-support mechanisms of photobiomodulation are theoretically relevant to both conditions. There is early research on LLLT for endometriosis-related pain, and the metabolic improvements seen with photobiomodulation may be relevant in PCOS given its mitochondrial and oxidative stress components. This is an area we are watching closely. We do not currently make condition-specific claims.
How long does it take to notice any effect? Cellular effects from photobiomodulation are measurable at the tissue level within sessions. Systemic effects - improvements in sleep, energy, cycle regularity - are typically reported within 3–6 weeks of consistent daily use. We recommend a minimum 12-week commitment when using the Kini for reproductive health support.
Does the Kini replace fertility treatment? No. Unambiguously, no. The Kini is a cellular support tool, not a medical device for the treatment of infertility. It is designed to be used alongside, and never instead of, appropriate medical care. If you are experiencing fertility challenges, please work with a qualified medical professional.
Which device is right for me? The Kini is the device best suited for fertility and hormonal support applications due to its targeted, wearable format and abdominal coverage. Our quiz at luminouslabs.health can help you identify whether the Kini or another device in the CellLight™ range is the right starting point for your goals.
8. Who This Article Is For
If fertility support is something you are actively exploring - whether you are preparing for IVF, navigating unexplained infertility, managing PCOS or endometriosis, or simply thinking about your reproductive health as part of a longer-term wellness approach - I wrote this for you.
I know how much noise there is in this space. I know how many products make claims they cannot support. I know how emotionally exhausting it is to separate signal from hope.
My commitment, with everything we publish at Luminous Labs, is to tell you exactly what we know, exactly what we don't, and exactly where the line is between good science and wishful thinking.
What we know: the cellular mechanism is sound. The existing peer-reviewed research is encouraging. Our clinical partnership is producing observations worth continuing to study.
What we don't know yet: the size of the effect, which patient populations benefit most, and the optimal protocol variables. That work is ongoing.
If you want to follow this research as it develops, subscribe to our newsletter. And if you have questions - about the science, the protocol, or whether the Kini makes sense for your situation - reply to any of our emails or reach out directly. We read every message.
References
¹ Bentov Y, Casper RF. (2013). The aging oocyte — can mitochondrial function be improved? Fertility and Sterility, 99(1), 18–22.
² Kinoshita J, Barbosa E. (2016). Low level laser therapy (LLLT) associated to IVF. Laser Therapy, 25(3), 175–182.
³ Hamblin MR. (2017). Mechanisms and mitochondrial redox signaling in photobiomodulation. Photochemistry and Photobiology, 94(2), 199–212.
⁴ Agarwal A, Aponte-Mellado A, Premkumar BJ, Shaman A, Gupta S. (2012). The effects of oxidative stress on female reproduction: a review. Reproductive Biology and Endocrinology, 10(1), 49.
This article is for informational purposes only and does not constitute medical advice. If you are experiencing fertility challenges or are undergoing fertility treatment, please consult a qualified medical professional before adding any supplementary protocol to your care plan.












